Description
Magnesium Sulfate Heptahydrate
Magnesium Sulfate Heptahydrate
Magnesium Sulfate Heptahydrate Detail Show :
Core advantages:
High purity: State-of-the-art materials and techniques are applied during the manufacturing processes to ensure Product Purity ≥99.8%, with the Impurity level
Stable quality: The principles of the ISO9001 quality management system have to be applied in every instance where production takes place. This ensures 36 tests are performed on the products of each series for their stability. ”
Uniform particles/fine powder: Different sizes of the particles and/or powders are availabe apon customers’ or market demands to achieve a uniform distribution and enhance the reactivity of the product. ”
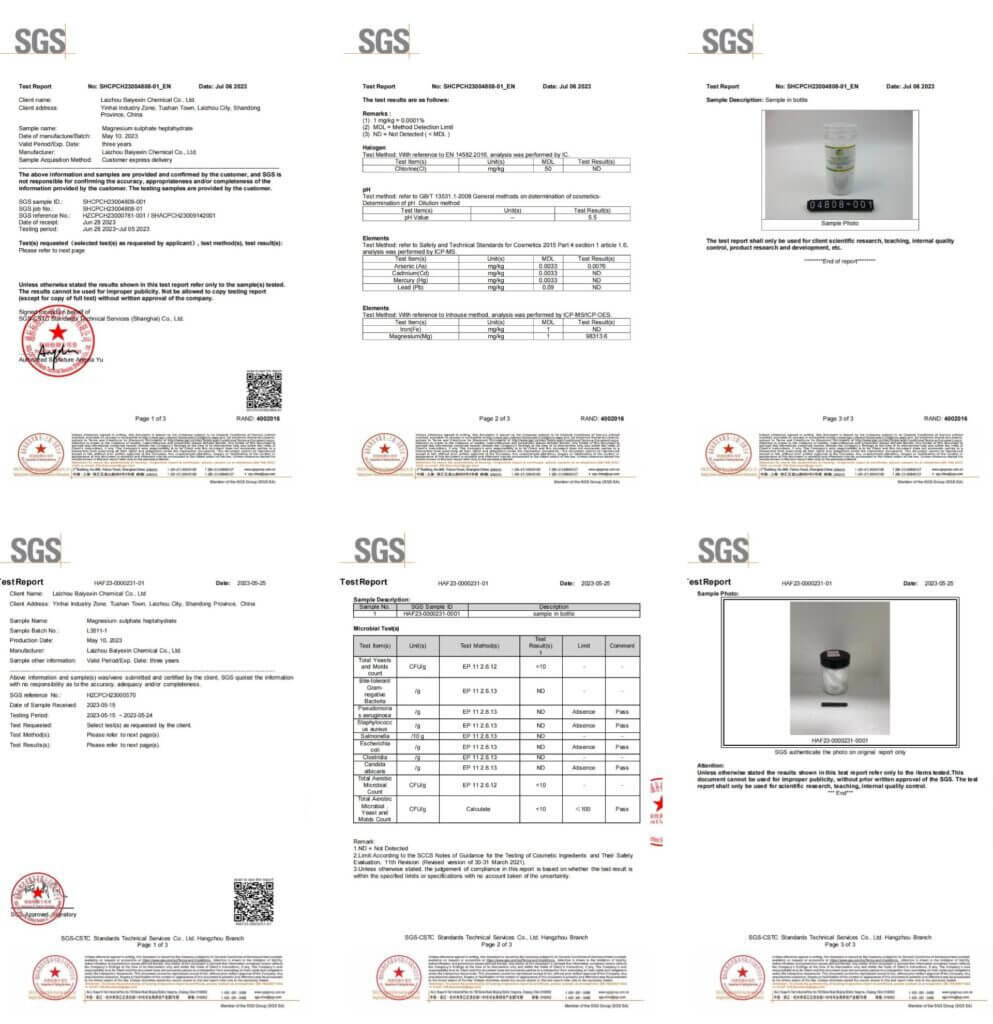
Magnesium Sulfate Heptahydrate Examining Report:
Packaging options offered by Baiyexin:
The company offers various product packaging options which include woven bags and PE bags and laminated bags and big bags while providing five different weight ranges which include 25kg and 50kg and 600kg and 1000kg and 1200kg.
Certification:

Factory Picture:
Laizhou Baiyexin Chemical Co., Ltd. is a top maker of Magnesium Sulfate Heptahydrate products. They focus on high purity and strict quality checks. Baiyexin provides custom solutions for different industries, making it a trusted name worldwide.

Overview
It is not wrong to say that magnesium sulfate heptahydrate or better known as Epsom salt is a very important inorganic salt with the chemically inherent formula of MgSO₄·7H₂O, and molecular mass equal to 246.47 g/mol. IUPAC chemical designations include integers CAS No. 10034-99-8 and EINECS No. 231-298-2. It is actively applied in such areas as agricultural production, the pharmaceutical field, industry, medicine, and everyday life, which involves, among others, mineral supplementation and manufacturing. It is due to its impressive compound’s stability, which allows one to use it for various purposes, and in turn is one of the most widely used magnesium salt set in the history of its production. The proposed article is intended to discuss magnesium sulfate heptahydrate considering all aspects in order to conduct a chemical review of the compound to its current stage in industrial applications with emphasis on some products and their production, purchase, and usage.
Key Takeaways
- Basic Information on Magnesium Sulfate Heptahydrate.
- Preparation method of magnesium sulfate heptahydrate.
- Application of magnesium sulfate heptahydrate.
- Magnesium Sulfate Heptahydrate Safety and Storage.
- Magnesium sulfate heptahydrate market and price.
- Laizhou Baiyexin Chemical.
1. Basic Information on Magnesium Sulfate Heptahydrate
1.1 Chemical Properties
- Chemical Name: Magnesium Sulfate Heptahydrate
- Aliases: Bitter salt, magnesium sulfate heptahydrate
- Magnesium Sulfate Heptahydrate Chemical Formula: MgSO₄·7H₂O
- Magnesium Sulfate Heptahydrate Molecular Weight: 246.47 g/mol (of the salt MgSO₄ * 7H₂O; MgSO₄ = 120.37 g/mol and 7 H₂O = 126.10 g/mol)
- Structure: Contains a Mg²⁺ cation, a SO₄²⁻ anion, and seven water molecules, including one crystal water with the monoclinic symmetry. Sulphate ions are surrounded by water molecules and are determined by hydrogen bonding.
- Inorganic Salt Properties: It is a neutral salt of a strong acid (sulfuric acid) and a strong base (magnesium oxide)). When dissolved in water it will dissociate and break down into Mg²+, and SO₄²− ions. It is an inorganic compound that does not contain any of the organic functional groups in its structure.
1.2 Physical Properties
- Magnesium Sulfate Heptahydrate Appearance: White crystals that resemble needles or columns that are odorless with slightly bitter taste. At room temperature, water content as an isomer appears on the surface of a fine light powder with powder sizes 0.1–1 mm, 1–3 mm or 2–4 mm.
- Magnesium Sulfate Heptahydrate Solubility: Water (at about 35.1g per 100 ml at 20 degrees Celsius, dissolve endothermically) – very soluble; alcohols – poorly soluble (less than 0.5g in 100 ml) ; organic compounds such as acetone and ether are not soluble.
- Magnesium Sulfate Heptahydrate Density: 1.68 g ml³ (crystalline shape hydration, at temperature of 20°C).
- Key Physicochemical Parameters:Crystallization water starts to leave the crystal above 50 – 70°C, hence a melting point is absent. An anhydrous MgSO₄ melts at 1124°C. Nevertheless, at this temperature it melts and disintegrates. The boiling point of the substance is not considered as this compound sublimes on heating into MgO, SO₃ etc.
- Valency: Valency: +2 for Mg, +6 for S, and -2 for O. A preponderance of 1 mole of magnesium sulfate (MgSO₄) is found together with 7 moles of water (H₂O) in each mole of the MgSO₄. 7H₂O compound indicating that the substance is typical ionic in nature.
- Composition: 9.86% Mg, 13.01% S, 51.21% O, and 36.13% H₂O approximately. The content of pure MgSO₄ approximately comprises 48.8% and the hydrated moisture 51.2%.Composition: 9.86% Mg, 13.01% S, 51.21% O, and 36.13% H₂O approximately. The content of pure MgSO₄ approximately comprises 48.8% and the hydrated moisture 51.2%.
1.3 Chemical Stability Of Magnesium Sulfate Heptahydrate
- Room Temperature Stability: Table in normal conditions is non-oxidizable or does not decompose. In dry conditions it may lose its crystal water by 'efflorescence' i.e., converting to monohydrate or anhydrous magnesium sulphate, which appears as a white powder. When there is moisture available, the dehydrated forms can take up water again making it useful for desiccant and humidity control purposes.
- Temperature-Dependent Behavior:
- At temperatures between 50–70°C one water molecule is driven off from MgSO₄·7H₂O, resulting in MgSO₄·6H₂O.
- When heated between 70–100°C, it drives off five more water molecules and becomes MgSO₄·H₂O (monohydrate).
- Eventually, between 200 and 250°C, the final water molecule is removed to transform MgSO₄ into anhydrous magnesium sulfate.
- Anhydrous magnesium sulfate turns into magnesium oxide, sulfur (III) oxide, sulfur (IV) oxide, etc. at 600°C.
- Chemical Reactivity: Relatively stable, not prone to violent reactions.
- Using Bases: An example of a base that will react with MgBr₂ will be NaOH or KOH due to actual formation of Mg(OH)₂ being observed (e.g. MgSO₄ + 2NaOH → Mg(OH)₂↓ + Na₂SO₄).
- Using Salts: Combining MgBr₂ with BaCl₂ yields BaSO₄ precipitate, usedto find out presence of sulfate ions or with carbonates to give MgCO₃ precipitate.
- Using Acids: When hydrochloric acid and magnesium sulfate react, magnesium chloride is formed and sulfuric acid is also formed.
1.4 Differences and Transformations Among Hydrate Forms
Magnesium sulphate can be found in several hydrate forms of dodecahydrate, heptahydrate, hexahydrate, monohydrate and anhydrous which can interchange in different conditions of temperature and concentration and increasing complications in the formation of phase equilibria.
Table: Key Differences Among Magnesium Sulfate Hydrates
| Hydrate Form | Crystal Water | Solubility | Stability | Stable Temperature Range | Applications |
|---|---|---|---|---|---|
| Anhydrous (MgSO₄) | 0 | Highly soluble (exothermic) | Very stable (moisture-resistant) | >200°C | Desiccant, industrial additive |
| Monohydrate (MgSO₄·H₂O) | 1 | Moderately soluble | Stable (less prone to efflorescence) | >67.5°C | Industrial filler, feed additive |
| Hexahydrate (MgSO₄·6H₂O) | 6 | Moderately soluble | Prone to efflorescence | 48.1–67.5°C | Specialty industrial processes |
| Heptahydrate (MgSO₄·7H₂O) | 7 | Highly soluble (fast dissolution) | Prone to efflorescence | -1.8–48.18°C | Agricultural fertilizer, pharmaceuticals, bath salts |
| Dodecahydrate (MgSO₄·12H₂O) | 12 | Extremely soluble | Highly prone to efflorescence | -3.9–1.8°C | Specialty low-temperature applications |
Magnesium Sulfate Heptahydrate vs. Anhydrous:
- The maximum difference between both types of using of practice is that it occurs within the content of crystalline water as well as their properties. The heptahydrate type contains about 51% water bound in its crystal structure, while the magnesium sulphate anhydrous does not contain any crystal water. They differ in such properties as solubility, stability, as well as considerations in uses. Heptahydrate is not amorphous and is not hygroscopic therefore it is more convenient to determine its amount in industrial application; while anhydrous magnesium sulphate is hygroscopic therefor can be used particularly as a drying agent.
Transformation Relationships (text representation):
- Magnesite (MgCO₃) → Heat → Magnesium oxide (MgO)
- MgO + H₂SO₄ → Anhydrous MgSO₄
- Anhydrous MgSO₄ + H₂O → MgSO₄·7H₂O
- MgSO₄·7H₂O → Heat (120°C) → MgSO₄·H₂O
- MgSO₄·H₂O → Heat (250°C) → Anhydrous MgSO₄
- Anhydrous MgSO₄ → High heat (>1124°C) → MgO + SO₃
2. Preparation Methods Of Magnesium Sulfate Heptahydrate
Industrial Preparation: Main Processes and Applications
Industrial production focuses on resource utilization and cost control, with three primary methods:
Magnesite Acidolysis Method
- Raw Materials: Magnesite (MgCO₃ ≥85%, CaO <5%), industrial-grade sulfuric acid (H₂SO₄ 93%–98%).
- Steps: Crush magnesite → React with sulfuric acid (MgCO₃ + H₂SO₄ + 6H₂O → MgSO₄·7H₂O + CO₂↑) → Filter impurities (e.g., CaSO₄, Fe₂O₃) → Concentrate (evaporate to 35%–40%) → Cool and crystallize (20–30°C) → Centrifuge → Dry (50°C to avoid water loss).
- Applications: Suitable for magnesite-rich areas (e.g., Liaoning and Shandong, China). Produces 98%–99% purity products for agricultural and industrial use.
The direct processing of Epsomite Ore
- Ingredients: Epsomite ore (natural MgSO₄·7H₂O, ≥90%)
- Procedures: Ore mining → Crushing → Leaching with water (soluble impurities dissolution) → Filtration → Concentration and crystallization → Purification.
- Especially well uses in the natural mineral-containing location s (for example Qinghai Salt Lake, China; Utah, USA). Inexpensive and of simple technology, without creating pollution and other such costly auditing; some modifications may allow for up to >99% purity for food and pharma grades.
Byproduct Recovery Method
- Primary Substances: Saline spor with (Mg²⁺, SO₄²⁻ ions), steel plant pickling waste or used in coal power generation: a mixture of Mg and SO₄ containing waste.
- Operative Procedures: Saline solutions/effluents prepared from Mg containing brine/waste (removal of Ca, Na, and heavy metal ions) → solvents stripped off by evaporating means and surplus saline solution’s brine → NaCl and MgSO₄. 7H₂O slowly → Dry.
- Uses: Such is commonly employed in areas such as chemical parks and those situating lakes of salt. Is green; ratios salient, 97–99% pure. Moreover, the fact that the required inputs are used in ‘waste-to-resource’ manner significantly reduces the costs to 15–20% compared to the existing magnesite technology.
Laboratory Preparation: Small-Scale Synthesis
- Method: The Reaction of Magnesium Oxide with Sulfuric Acid
- Reagents: Minimum-purity (99% or higher) MgO, and pre-diluted sulfuric acid (1:3 to lower the risk of exothermic and in some cases explosive reaction).
- Steps: Fill the beaker with distilled water → Think of adding H₂SO₄ very slowly → Start putting some quantities of MgO till about pH 6 to 7 is achieved and the solution does not produce gas → Raise temperature to fundamental 50°C and stir → Separate unreacted MgO by filtering → Concentrate until a layer of dissolved substance forms → Set aside for a day in a cool place → Slake and isolate MgSO₄·7H₂O which is in crystals (>99.5%).
- Reaction: MgO + H₂SO₄ + 6H₂O = MgSO₄·7H₂O.
- Key Details: The speed of addition of H₂SO₄ should be very strict not to allow unwanted heat release, additionally it should not go above the given temperature of 50°C as the dissipated temperature affects the water of crystallization and Washings must be performed with cold water in order to reduce the amount of impurities in the crystals.
Process Comparison and Dehydration Reactions
Table: Comparison of Preparation Methods
| Method | Purity | Crystal Form | Relative Cost | Environmental Impact |
|---|---|---|---|---|
| Magnesite Acidolysis | 98%–99% | Needle-like crystals | 100 | Moderate (CO₂ emissions) |
| Epsomite Ore Processing | 99%–99.5% | Columnar crystals | 80 | High (no wastewater) |
| Byproduct Recovery | 97%–99% | Powder/granules | 70 | High (wastewater recycling) |
Dehydration Reactions:
- MgSO₄·7H₂O (s) → MgSO₄·H₂O (s) + 6H₂O (g) (ΔH = +126 kJ/mol, exothermic)
- MgSO₄·H₂O (s) → MgSO₄ (s) + H₂O (g) (ΔH = +48 kJ/mol, exothermic)
- Total: MgSO₄·7H₂O (s) → MgSO₄ (s) + 7H₂O (g) (ΔH = +174 kJ/mol, endothermic)
Natural Sources and Derivatives
- Natural Form: It is naturally found as the mineral epsomite (MgSO₄·7H₂O) from saline lakes and deposits of minerals like Chaka Salt Lake in Qinghai, China or Solikamsk in Russia. High-quality epsomite is readily available and can be used without further purification.
- Derivatives:Other aged additives encompass potassium sulfate solution containing magnesium sulfate (K₂SO₄·MgSO₄·6H₂O) applied to plants used for compound fertilizer and ammonium sulfate with magnesium sulfate ((NH₄)₂SO₄·MgSO₄·6H₂O) overwatered or applied directly for agricultural top-dressing; all are prepared through metathesis utilizing salts.
Byproduct Management and Environmental Requirements
- Byproducts: Magnesite method produces CO₂ (recyclable for dry ice or sodium bicarbonate), acidolysis produces waste slag (CaSO₄, Fe₂O₃, used in construction), and wastewater (low Mg²⁺, neutralized to pH 6–9 before discharge).
- Environmental Standards: In China, complies with GB 8978-1996 (wastewater COD <100 mg/L, heavy metals <0.01 mg/L). EU REACH regulations require no persistent organic pollutant emissions.
3. Applications Of Magnesium Sulfate Heptahydrate
3.1 Agriculture: Magnesium and Sulfur Fertilizer, Feed Additive
- Primary Use: Two-component fertilizer source providing magnesium and sulphur nutrients.
- Mechanism: Magnesium forms the basis of chlorophyll which drives the process of photosynthesis. Sulfur provides support in the formation of amino acids and enzymes thus enhancing the quality of crops (e.g fruit sugar levels).
- Applicable Soils: Soils deficient in magnesium (e.g. acidic red soils or sandy soils).
- Suitable Crops: Citrus fruits, grapes, tomatoes, rice, soybeans, potatos, roses and significantly increases leafy vegetables yields.
- Application Methods: Foliar application (0.2% solution - 0.5% solution), soil application (5 kg - 20 kg per acre).
- Precautions: Use appropriate quantity applicable to the crop and soil conditions, preferably with the help of recommendations after carrying out a soil test. Excessive application may result in salt injury and nutrient discrepancy.
Comparison with Other Magnesium Sulfate Fertilizers:
| Fertilizer | Solubility | Soil pH Impact | Absorption Efficiency | Relative Cost |
|---|---|---|---|---|
| MgSO₄·7H₂O | High | Neutral | 85%–90% | 100 |
| MgO | Low | Alkaline (raises pH) | 40%–50% | 80 |
| MgCl₂ | High | Neutral | 75%–80% | 90 |
Advantages of Magnesium Sulfate Heptahydrate:
- High solubility enhances the absorption of nutrients by plants.
- Quick and effective in countering a magnesium deficient effect in crops.
- It provides plants with both magnesium and sulfur.
- Is gentle to the soil, stable, and does not change the soil pH.
Feed Additive:
- Function: Helps the livestock (e.g. swine, fowls, cattle, horses) to provide magnesium and improve meat quality, prevent stress syndrome such as muscle spasm, nervous excitement or constipation and boost defense mechanisms.
- Standards: Meets the requirements of China National standard GB/T 26451-2011 as general purpose specification requirement for MgSO4*7H2O, Magnesium sulfate 7-Hydrate (purity ≥98%, lead content ≤0.001%).
- Application: Use mixed with feed for horses 10–20 g/day.
3.2 Pharmaceuticals
In the medical domain, magnesium sulfate heptahydrate is often applied as a magnesium additive and as a therapeutic agent because of its “mild effects and multiple administration routes”.
- Laxative: It involves oral administration which enhances osmotic pressure in the intestine and retains water resulting in stool softness and induces peristalsis. This is utilized for treating constipation and prior to bowel examination.
- Choleretic: It acts on the mucus membrane of the duodenum making the gallbladder contract and secrete bile for cases of cholecystitis and gallstones.
- Anticonvulsant: In the case of intra-venous administration, it reduces the activity of the central nervous system to that extent. This is meant for cases of eclampsia and convulsions due to tetanus.
- Anti-inflammatory (Topical): A 50 % diluted solution is used for wet dressings in the management of inflammatory edema and phlebitic veins.
Table: Pharmaceutical Applications
| Administration | Indications | Mechanism | Typical Dosage/Concentration |
|---|---|---|---|
| Oral | Constipation, bowel prep | Increases osmotic pressure | 5–10 g/dose, max 30 g/day |
| Oral | Cholecystitis, gallstones | Stimulates bile secretion | 2–5 g/dose, diluted |
| IV Injection | Eclampsia, anticonvulsant, hypomagnesemia | Suppresses neuromuscular excitability | 2.5–4 g initial dose, slow injection (>10 min) |
| Topical | Soft tissue swelling (phlebitis, burns) | Osmotic dehydration | 50% solution, 2–3 times/day |
Common Products:
- Oral: it is available as Magnesium sulphate in solution (10%) and a powder (5 g/sachet).
- Injection: This is in a 50% concentration of approximately 1 g/ml; include the manufacturer (e.g. Huabei Pharma, Shijiazhuang Pharma, Shanghai Pharma; 20 ml contains 10 g in solution).
- Topical agents come as 33% and 50% solutions.
Contraindications: Advanced stages of renal dysfunction (creatinine clearance
Side Effects: The use of an overdose may cause loose stools, and dehydration (in case the drug was administered orally); low blood pressure, depression of breathing (in case of IV administration); skin irritation (if used topically for an extended period).
3.3 Industrial Applications of Magnesium Sulfate Heptahydrate
- Paper Industry: employed as a sizing agent in combination with rosin with the purpose of enhancing the water resistance of paper as well as increasing the whiteness and smoothness of paper by way of filling it. Consistent with QB/T 4446-2012.
- Textile Dyeing: Functions as a mordant in the dyeing of cotton or linen to improve the adhesion of dyestuffs to the fiber, as well as a fixing agent to enhance the chromatic intensity.
- Flame Retardants: They are used for the construction of cables and some building materials when combined with either PVC, acrylics, epoxy or polyesters. These chemicals decompose and liberate the water of crystallization that can dissipate heat as well as decomposing to MgO which in turn is oxygen retarding.
- Coatings: Serves as a pigment extender which provides better flow and scrub resistance in latex paints, and also as an anti-corrosion pigment used in metal primers.
- Food Additives: Meets GB 25588-2010 requirement (≥99.0% for MgSO₄ and ≤0.0002% for As) standards. Utilised for stabilisation of icecream or a dairy such as milk, used as a coagulation agent in tofu. Three to seven grams per one kilogram for dairy and one point four to two point eight grams per kilogram for beverages and zero point zero five grams per one kilogram for mineral drinks.
- Leather Industry: These supplements are meant for filling, strengthening and also for leather enhancement.
- Construction: Additions of these materials strengthen and enhance fire property of cement based and gypsum based products and composites.
- Electroplating: Is entered into the plating systems as a conductive salt or also as the additive for coating improvement.
3.4 Daily Life and Other Uses
- Bath/Foot Soak Rate: Bath/Foot Soak: Utilize joints and in a 5%-10% solution, it can be safely used to wash the body, moisturizing the back of the bath, and energizing the feet.
- Skincare: Solutions of 33% help in the treatment of acne and eczema, as well as being ingredients in cosmetic products functioning as exfoliators or oil control agents.
- Cosmetics: It has also been used in creams and masks to help humectify and repair the layers of skin or as a haircare product for people with oily skin.
- Water Treatment: Precipitates calcium and/or magnesium and further can be used for softening the water as well as removal of calcium and magnesium and also phosphate ions.
- Research: Used as an analytical reagent – e.g. for EDTA Method of Titration or as a component of culture media – the source of magnesium in media for the growth of microorganisms. The purity must be not less than 99.5%.
4. Comparisons and Relationships
4.1 Differences with Common Salts
Magnesium Sulfate Heptahydrate vs Sodium Carbonate:
| Property | Magnesium Sulfate Heptahydrate | Sodium Carbonate |
|---|---|---|
| Formula | MgSO₄·7H₂O | Na₂CO₃ |
| pH | Neutral (6.5–7.5) | Alkaline (11–12) |
| Uses | Fertilizer, pharma, flame retardant | Detergent, glass, food additive |
| Reactions | Forms Mg(OH)₂ with bases | Forms CO₂ with acids |
Magnesium Sulfate Heptahydrate VS Magnesium Chloride Hexahydrate:
| Property | Magnesium Sulfate Heptahydrate | Magnesium Chloride Hexahydrate |
|---|---|---|
| Anion | SO₄²⁻ | Cl⁻ |
| Corrosivity | Low (non-corrosive) | High (corrosive to iron/steel) |
| Uses | Agriculture, pharma, cosmetics | Construction (magnesite boards), snow melting |
| Environmental Impact | No chlorine, soil-friendly | Contains chlorine, may cause soil compaction |
Magnesium Sulfate Heptahydrate or Epsom Salt: This is the Same Thing; "Epsom Salt" is the Appellative, brought forth out of the discovery of it in Epsom, England and that is facilitates bowel movement.
Magnesium Sulfate versus Magnesium Sulfate Heptahydrate: "Magnesium sulfate" is an inclusive term touching upon all hydrate configurations; septyhdrate is the main type used in manufacturing.
4.2 Comparison with Other Magnesium Sulfate Fertilizers
See Section 3.1. Main advantages: high solubility, rapid absorption, neutral pH. Disadvantages: higher cost, prone to leakage (requires multiple applications).
4.3 Environmental Comparison (vs. Zinc Sulfate, Calcium Sulfate)
- Production: Magnesium sulfate heptahydrate is used as a byproduct, hence its production is less costly to the environment than zinc sulfate (permits oil mining) and calcium sulfate (permits gypsum mining).
- Post-Use Impact: Unlike CaSO₄ that causes every farmer’s nightmare that is soil compaction or ZnSO₄ with heavy metals that biomagnify, MgSO₄·7H₂O is completely absorbed by the plant without a trace.
- Waste Disposal: On the other hand, MgSO₄·7H₂O and its other forms are naturally degraded by water, unlike ZnSO₄ and CaSO₄ which need to undergo sophisticated waste disposal mechanisms.
5. Safety and Storage of Magnesium Sulfate Heptahydrate
5.1 Safety and Hazard Identification
- Classification: It is not regarded as a hazardous chemical and as such, is not on the 2022 China Directory. However, mild irritation may occur when exposed to high levels of it, which is why it is viewed as a general chemical.
- Toxicity: The substance is not very toxic (the LD50 for the rat as per oral administration is 6450mg/kg). Dust inhalation for a long time may lead to respiratory tract irritation, solutions with high concentration may cause skin irritation in the form of slight redness to the skin, and excess intake (>50g) may induce vomiting and electrolytes disturbance.
- Hazard Levels: In the first instance, the irritation from ingestion or inhalation is mild from a clinical point of view.
5.2 First Aid
- Skin Contact: The user is to remove any clothes that may carry the contamination and wash the skin for fifteen minutes. Should there be any inflammatory responses, calamine can help.
- Inhalation: Transport to an open area, clarify that airways are open. Observation for cough or shortness of breath is important as they can warrant medical help.
- Ingestion: Encourage vomiting with the administration of warm water in the case of a conscious patient, and go to the hospital while carrying the product. There should be no use of antidiarrhea medicines.
5.3 Handling and Storage
- Protective Measures: Nitrile gloves, N95 masks, and goggles must be worn. Air ventilation should be maintained at 0.5 m/s or higher avoid dust clogging & other airborne filth. Hygiene is also key, and therefore hands or face should also be washed after all processes.
- Storage Conditions:
- Keep inside a cool and dry location where the temperature does not exceed 30°C and the humidity remains within 60% without sunlight exposure
- Utilize PE or PP drums, or earthenware jars while excluding iron containers (sulfate corrosion).
- Storage should be away from high pH substances like sodium hydroxide or sodium carbonate and or oxidizers such as potassium permanganate at a distance of 1 meter and more.
- Standardization period: 1-2 years (for agricultural/industrial uses), 2-3 years (for food/medicine). Please make top up within 3 months of opening in order to void the bloom.
5.4 Emergency and Waste Handling
- Spills: Use plastic brooms. Particularly, solid waste should be put in sealed bags. Liquid releases must be absorbed with sand, neutral followed by water such that pH is adjusted to 6.0-9.0.
- Fire/Explosion: The product will not burn or it will not explo- de. Extinguish fires in the vicinity by means of water and remove the containers away so as not to produce any dust.
- Waste Disposal: Adjust the pH of the wastewater in the range of 6–9 before settling and disposing of the wastewater (GB 8978-1996). Agricultural or industrial grade waste is useful for soil improvement, while grades used in pharmaceuticals or food must be disposed of as clinical waste.
6. Magnesium Sulfate Heptahydrate Market And Development
6.1 Market Size and Drivers
- Global Market (2025): Approximated at 1.5 million metric tons per annum. Principal areas include the following countries: China 50 percent, North America 20 percent, Europe 15 percent, rest 9 percent.
- Application Breakdown: 55 percent agriculture, industry 30 percent, 10 percent pharmaceuticals and 5% other industries.
- China Market: It has a capacity of up to 750 thousand tons and Shandong province constitutes 35%, Qinghai 25% and Jiangsu 15%.
- Drivers:
- Agriculture: The increased circulation of regional expansion with magnesium-depleted soils (e.g. red soil of South China regions) and intensification in the commercial farming in the production of respective crops (e.g. citrus, grapes and vegetables).
- Pharmaceuticals: There is minimal fluctuation in the requirement for therapies aimed at eclampsia treatment and there is also an increase in occurrence of alienated magnesium deficiency in patients.
- Industry: Under the EU requirements set out in RoHS version 2.0 directive, brominated flame retardants are to be replaced with inorganic flame retardants.
6.2 Major Producers
| Company | Region | Capacity (tons/year) | Purity Range | Competitive Edge |
|---|---|---|---|---|
| Laizhou Baiyexin Chemical | Shandong, China | 300,000 | 98%–99.5% | High cost of production, low quality work [efficiency], low cost factory (cheap labor) and flexible pricing |
| PQ Corporation | USA | 60,000 | 99%–99.8% | Cutting edge technology, premium pharmaceuticals |
| Giles Chemical | USA | 50,000 | 98.5%–99.5% | Well mapped overseas markets, an industrial orientation in the production scale |
| Laizhou Laiyu Chemical | Shandong, China | 120,000 | 98%–99.5% | Rich and much exploitable resources, high purity |
| K+S Group | Germany | 40,000 | 98%–99.5% | Distributive network, European market share, accent on agriculture |
6.3 Trends and Opportunities/Challenges
- Trends:
- Green Production: Encourage recovery of by-products, and use of low-temperatures for crystallization processes (with an energy saving amount of 30%).
- High-Value Products: Pharmaceuticals of the highest purity (>99.8%), cosmetics in nanometer size, slow-releasing fertilizers.
- Environmental Tech: Remediation of wastewater with no emissions, there is also sufficient recycling of slags into construction.
- Opportunities:
- Emerging uses: Additives for the lithium battery electrolyte, drug delivery for magnesium which will only reach the target area.
- Policy support: Green inputs are pushed forward by rural development goals in China and Green Deal of the European Union.
- Challenges:
- Cost fluctuations: Prices of raw materials (such as sulfuric acid and magnesite) fall within a range of 20%–30% of their current prices.
- Environmental regulations: With China’s focus on carbon emissions, the country might not favor energy-intensive procedures.
- Market competition: With the homogenization of low-purity products, there is a decline in profits.
7. Market Price Of Magnesium Sulfate Heptahydrate And Its Influencing Factors
Table: Market Prices by Purity (2025, China)
| Grade | Purity | Price (CNY/ton) | Packaging |
|---|---|---|---|
| Agricultural | 98% | 500–700 | 25 kg/50 kg bags, 1000 kg bulk |
| Industrial | 99% | 550–750 | 25 kg/1000 kg bags |
| Food | 99.2% | 2000–2500 | 25 kg sealed drums |
| Pharmaceutical | 99.5% | 3000–5000 | 1 kg/5 kg aluminum-plastic bags |
| Laboratory | 99.8% | - | 500 g/1 kg reagent bottles |
- Price Factors:
- Raw Materials: The increase in the prices of sulfuric acid or magnesium oxide could in turn bid the prices up by about 10% up to 15%. For instance a unit of sulphuric acid which was previously at 500 CNY is now at 800 CNY.
- Supply/Demand: The spring season allowing crops planting and increase in demand; therefore, an increase of 5%–8% in prices occurs.
- Purity/Specification: There is a hike in prices to the extent of 10% to 15% in cases of 0.5% better purity, and 10% to 15% in the case of granules than powder.
- Transport: Distant locations (South China, for instance) mean an additional expense of 200-300 CNY/ton.
- Policy: Regulations regarding environment suggest a possible price increase of around 20 to 30 percent.
8. Magnesium Sulfate Heptahydrate Industry Standard
8.1 Pharmacopoeia Standards
| Pharmacopoeia | Region | Purity (MgSO₄) | Water Content | Heavy Metals (Pb) | Arsenic (As) |
|---|---|---|---|---|---|
| USP | USA | ≥99.0% | 13.0%–16.0% | ≤0.001% | ≤0.0002% |
| EP | Europe | ≥99.0% | 13.0%–16.0% | ≤0.001% | ≤0.0001% |
| ChP | China | 99.5%–100.5% | 13.0%–16.0% | ≤0.001% | ≤0.0002% |
| IP | India | ≥99.0% | 12.5%–16.5% | ≤0.001% | ≤0.0003% |
| BP | UK | ≥99.0% | 13.0%–16.0% | ≤0.001% | ≤0.0002% |
8.2 Industrial Standards
- China GB Standards:
- GB/T 26568-2011 (Industrial): Agricultural ≥98.0%, water-insoluble ≤0.2%, pH 5.0–9.0; Industrial ≥99.0%, water-insoluble ≤0.1%.
- GB/T 26451-2011 (Feed): MgSO₄·7H₂O ≥98.0%, Pb ≤0.001%, As ≤0.0002%.
- GB 25588-2010 (Food): MgSO₄ ≥99.0%, Pb ≤0.0005%, Hg ≤0.0001%.
- International: ISO 8298-2000: Industrial MgSO₄ ≥98.0%, water-insoluble ≤0.5%.
8.3 EU REACH Compliance
- Registration: It is to be done if the production is equal to or more than 10 tons in a year ( for example, registration number 01-2119584257-46-0000).
- Restrictions: the product should not contain any heavy metal, ( Cd, Hg) more than 0.0001% and shouldn’t contain phthalates.
- Notification: It is to be done when the substance is for cosmetic or pharmaceutical use to ECHA.
- Testing: To prove absence of acute toxicity, skin corrosivity and degradation of the product, respectively i.e. OECD 301B.
9. Magnesium Sulfate Heptahydrate Purity Grades
9.1 By Use and Purity
| Grade | Purity (MgSO₄) | Impurities | Applications |
|---|---|---|---|
| Agricultural | ≥98.0% | Water-insoluble ≤0.2%, Ca ≤0.5% | Soil/leaf fertilizer |
| Industrial | ≥99.0% | Water-insoluble ≤0.1%, Fe ≤0.005% | Paper, dyeing, flame retardants |
| Food | ≥99.2% | Pb ≤0.0005%, As ≤0.0002% | Food stabilizer, coagulant |
| Pharmaceutical | ≥99.5% | Pb ≤0.0001%, bacteria ≤100 CFU/g | Oral, injectable, topical drugs |
| Laboratory (AR) | ≥99.5% | Total impurities ≤0.5%, Cl⁻ ≤0.005% | Chemical analysis, research |
| Extrapure | ≥99.8% | Total impurities ≤0.2%, metals ≤0.0001% | High-end research, biomedicine |
9.2 By Form and Particle Size
- Forms:
- Powder (<0.1 mm): Fast-dissolving, used in foliar fertilizers, pharma powders, lab reagents.
- Granules: Fine (0.1 to 1 mm), medium (1 to 3 mm), and coarse (2 to 4 mm). Excellent flow behavior, often mixed in soil-fertilizing agents and in industrial fillers.
- Applications:
- Magnesium Sulfate Heptahydrate 0.1–1 mm: Greenhouse crops.
- Magnesium Sulfate Heptahydrate 1–3 mm: Field crops (corn, rice).
- Magnesium Sulfate Heptahydrate 2–4 mm: Orchards (citrus, apples).
10. Identification And Quality Control Of Magnesium Sulfate Heptahydrate
10.1 Identification Methods
- Laboratory:
- Sulfate: Add barium chloride solution (BaCl₂) the formation of a white barium sulfate (BaSO₄) precipitate appears insoluble in hydrochloric acid (HCl).
- Magnesium: Additional NaOH reagent forms the magnesium hydroxide (Mg(OH)₂) precipitate which is not dissolved in sodium hydroxide in excess.
- Crystal Water: By increasing the temperature of one gram of the sample up to 200°C, about fifty-one percent of the mass is lost to cater for presence of hepta hydration.
- Field:
- Appearance: The crystals are white in color, needle or column shaped and taste slightly bitter.
- Solubility: It dissolves in water producing a clear solution without precipitates.
- pH: In a 5% solution, the pH will be 6.5-7.5 (neutral unlike Na₂CO₃ which is alkaline).
10.2 Common Impurities
- Calcium Salts (CaSO₄): Prevent absorption of magnesium, contribute to scale formation within process equipment.
- Iron Salts (Fe₂(SO₄)₃): Lead to the yellowing of pharmaceutical and garnishment products.
- Heavy Metals (Pb, Cd, Hg): Not acceptable for food and pharmaceuticals due to toxicity.
- Insoluble Matter (SiO₂): Loss of transparency of injections or cosmetics.
- Impact: Pharmaceutical> Food>Industrial>Agricultural Effectiveness ofthe restrictions decreases respectively on the use of different materials families containing active compounds.
10.3 Quality Standards and Procurement
- Standards: Refer to pharmacopoeias (pharma/food), GB/T 26568-2011 (industrial/agricultural).
- Key Metrics:
- Purity: not lower than 98% for agriculture, 99% for industry and 99.5% for pharmaceutical use.
- Impurities: lead no more than 0.001% for medi- / yummies and non-dissolving in water no more than 0.2% for agri-products.
- Water Content: limits 13%–16% (dry = efflorescence; wet = deliquescence).
- Particle Size: Choose based on use (powder for foliar, 1–3 mm for soil).
- Packaging: should remain in one piece, readable information on the package (contents, date of manufacturing, expiry date).
11. Magnesium Sulfate Heptahydrate Packaging Options
| Type | Material | Weight/Capacity | Applications |
|---|---|---|---|
| Small | PE reagent bottles | 500 g, 1 kg | Lab, small-scale pharma/food trials |
| Medium | PP woven bags | 25 kg, 50 kg | Agricultural/industrial transport |
| Large (Bulk) | PP bags + PE lining | 1000 kg, 1250 kg | Industrial bulk (paper, flame retardants) |
| Sealed | Aluminum-plastic bags, PE drums | 1 kg, 5 kg, 25 kg | Pharma/food (moisture-proof) |
| Bulk | Tank trucks (liquid), open trucks (solid) | 20–30 tons | Large-scale supply (salt lake enterprises) |
- Notes: Pharmaceutical and food containers have to follow safety standards for examples GB 4806 (plasticizers should not migrate and should not have any addition) so I suggest using sealed drums for long-term storage.
12. Laizhou Baiyexin Chemical
12.1 Quality Assurance and Certifications
- Certifications: ISO 9001, ISO 14001, ISO 45001, EU REACH, Halal, Kosher.
- Product Certifications: Pharmaceutical certifications include compliance with ChP 2020, USP 43 and EP 10, whereas food-grade compliance is 25588-2010. They have also undergone third party testing of which laboratories SGS and Intertek were used.
- Quality Control: The systems include testing of raw materials, validation every 2 hours during the process, complete testing of the final product. No such product is supplied if it doesn’t conform.
12.2 Competitive Advantages
- Technology: There is an advantage of using patented crystallization and dehydration under low temperatures (ZL 202210234567.8), which comes with 25% less energy consumption, uniformed crystals are obtained, 90% of the crystals are within the range of 1 to 3 mm.)
- Capacity: It provides 300,000lt/day production which not only makes it possible to fulfill large demands (>5000lt) creates but also enhances delivery in 7–10 days.
- Customization: We can produce various high-purity (98-99.8%) magnesium sulfate heptahydrate with different particle sizes (0.1-4 mm).
12.3 Production and Services
- Lines with automated DCS (total control), sufficiently robust batches (notably that variations of purity are not exceeding 0.2%), and a pharmaceutical-grade cleanroom with a class rating no less than 10,000 with individual design.
- Services:
- Pre-Sale: we shall be able to offer you samples, submit test reports and give guidance on application of product, for example when used as fertilizer and in industrial formulations.
- In-Sale: Tracing of logistics, preparation of export documents, and assistance in inspection of goods for customers.
- After-Sale: Allow up to 24 hours for responses and 48 hours for resolutions, in the event that any quality problems arise (returns or replacements).
12.4 Case Studies
- Agriculture: Sent Granules of Magnesium Sulfate Heptahydrate1–3 mm to the giant China National Chemical Corporation, for 200,000 acres of citrus gardens reducing magnesium deficiency from 35% to 8%, sweetening the fruits by 1–2 degrees.
- Pharma: Manufactured 50% injection solution for Huabei Pharma, using 99.5% pharmaceutical grade, hundred percent compliance with ChP 2020.
- Industry: Zhejiang Chuanhua (textile dyeing) – shipment of products with 99% finished grades, that helps to reduce the use of dyes in 15% respectively and increases the degree of color retention in one degree.
13. FAQs
Is Magnesium Sulfate Heptahydrate the same as Epsom salt?
Yes, they are identical. Epsom salt is the common name due to its laxative effect.
Is Magnesium Sulfate Heptahydrate the same as Magnesium Sulfate Heptahydrate?
No. Magnesium Sulfate Heptahydrate is a general term for all hydrate forms; heptahydrate is the most common.
Is Magnesium Sulfate Heptahydrate organic or inorganic?
Inorganic, lacking C-C or C-H bonds.
Is Magnesium Sulfate Heptahydrate hygroscopic?
Weakly hygroscopic but prone to efflorescence in dry conditions. Anhydrous Magnesium Sulfate is strongly hygroscopic.
Is Magnesium Sulfate Heptahydrate soluble in water or ethanol?
Highly soluble in water (35.1 g/100 mL at 20°C), slightly soluble in ethanol (<0.5 g/100 mL), insoluble in acetone.
What does Magnesium Sulfate Heptahydrate look like?
White needle/columnar crystals or fine white powder, odorless.
Is Magnesium Sulfate Heptahydrate acidic, basic, or neutral?
Neutral (pH 6.5–7.5 in 5% solution).
Is Magnesium Sulfate Heptahydrate ionic or covalent?
Ionic, formed by Mg²⁺ and SO₄²⁻ ions.
Is Magnesium Sulfate Heptahydrate safe for pregnant women?
Safe for eclampsia treatment (IV) under medical supervision; oral use (laxative) requires caution; topical use is safe.
Can Magnesium Sulfate Heptahydrate be used for bathing?
Yes, 50–100 g in warm water (38–40°C) for Epsom salt baths, 1–2 times/week.
What is the content of Magnesium Sulfate Heptahydrate?
~48.8% MgSO₄, ~51.2% crystal water. Agricultural-grade ≥98%, pharma-grade ≥99.5%.
Can Magnesium Sulfate Heptahydrate be used as foliar fertilizer?
Yes, use 0.2%–0.5% solution during growth stages (e.g., citrus fruiting) for fast magnesium uptake.
Magnesium Sulfate Heptahydrate 0.1-1mm-BAIYEXIN

BaiYeXin is the largest magnesium sulfate heptahydrate manufacturer in China, providing magnesium sulfate heptahydrate 0.1-1mm.
Product SKU: BYX40448
Product Brand: BAIYEXIN
Product Currency: USD
Product Price: 90
Price Valid Until: 2026-12-31
Product In-Stock: InStock
5








BaiYeXin –
Baiyexin's Magnesium Sulfate Heptahydrate (MgSO₄·7H₂O) 0.1-1mm has excellent flowability during granulation and does not clump, greatly reducing the risk of material blockage in our production line.