Description
Magnesium Sulfate Heptahydrate
Magnesium Sulfate Heptahydrate
Magnesium Sulfate Heptahydrate Detail Show :
Core advantages:
The company achieves high product purity through its advanced production technology which delivers purity levels exceeding 99.8% and impurity levels below 0.1%, thus guaranteeing reliable protection of your production and experimental work.
The company achieves stable product quality through its complete adherence to ISO9001 quality management system which requires 36 testing procedures for every product batch to verify consistent product performance.
The products which we offer come in different mesh sizes and particle sizes to meet your requirements for optimal dispersibility and reactivity during your applications.
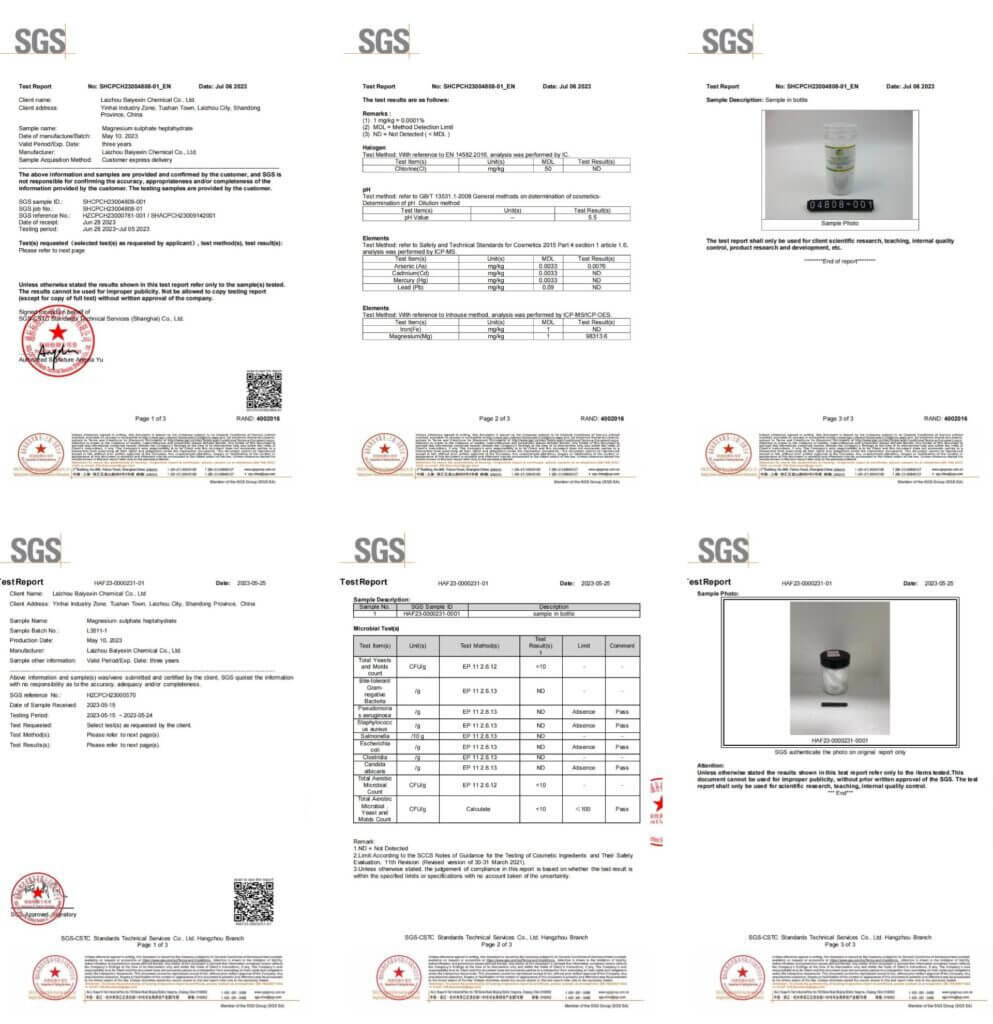
Magnesium Sulfate Heptahydrate Examining Report:
Packaging options offered by Baiyexin:
The company offers various product packaging options which include woven bags and PE bags and laminated bags and big bags while providing five different weight ranges which include 25kg and 50kg and 600kg and 1000kg and 1200kg.
Certification:

Factory Picture:
Laizhou Baiyexin Chemical Co., Ltd. is a top maker of magnesium sulfate products. The company emphasizes production of highly pure materials which undergo thorough quality assessment procedures. Baiyexin provides custom solutions for different industries, making it a trusted name worldwide.

Overview:
Magnesium Sulfate Heptahydrate serves as an essential inorganic salt compound. The compound displays the chemical formula MgSO₄·7H₂O and has a molecular weight of 246.47 grams per mole. The substance has been assigned a CAS Registry Number 10034-99-8 and an EINECS Number 231-298-2 for its international chemical identification. The material finds extensive application across different sectors because it possesses multiple uses as a nutritional supplement and industrial material and medical product. The product serves as a fundamental item within the magnesium salt product line because it maintains chemical stability and can be used in various situations which create high market demand. This article will present a complete research study about all key information regarding Magnesium Sulfate Heptahydrate which includes its fundamental properties and current industrial applications to assist readers with their needs for production and purchasing and practical use and other purposes.
Key Takeaways
- Basic Information on Magnesium Sulfate Heptahydrate.
- Preparation method of magnesium sulfate heptahydrate.
- Application of magnesium sulfate heptahydrate.
- Magnesium Sulfate Heptahydrate Safety and Storage.
- Magnesium sulfate heptahydrate market and price.
- Laizhou Baiyexin Chemical.
Product Overview
I. Basic Information of Magnesium Sulfate Heptahydrate
1.1 Chemical Properties
1.2 Physical Properties
- Appearance: At room temperature, it forms white acicular or columnar crystals that are odorless and with a slightly bitter taste; the powdered form appears to as a fine white powder. Granulated products with defined particle sizes such as magnesium sulfate heptahydrate 0.1-1mm, 1-3mm, 2-4mm are also exist.
- Solubility: Freely soluble in water (35.1g/100mL at 20℃ in water, heat is absorbed during solution); slightly soluble in ethanol
- Density: 1.68g/cm³ (measured at 20℃, crystals).
- Key Physicochemical Parameters: The heptahydrate starts to loose its water of crystallization when it is heated to 50-70℃; it does not have a specific “melting point” (it suddenly transforms into anhydrous magnesium sulphate at relatively high temperatures of 1124℃ melting point but not molten, it decomposes at melting; ); no boiling point (it is practically").
- Valence: Mg +2, S +6, O -2, this might be reflected by the structure of the compound where every one molecule of the heptahydrate contains one mole of MgSO₄ and seven moles of water, and this is very much an ionic compound.
- Composition: The mass percentage of Mg is approximately 9.86%, S is 13.01%, O is 51.01% and H₂O is 36.13%. In theory, the proportion of MgSO4 is roughly 48.8% and the remaining 51.2% is the water crystals.
1.3 Chemical Stability
Temperature Dependence:
The result of heating the water of crystallization is that it is lost in a stepwise manner:
- a. 50-70℃: Transformation of MgSO₄·7H₂O to MgSO₄·6H₂O (loses 1 molecule of water);
- b. 70-100℃: Further loses 5 molecules of water and a magnesium sulfate monohydrate (MgSO₄·H₂O) is created;
- c. 200-250℃: It then follows that the remaining 1 molecule of water is removed resulting in anhydrous magnesium sulfate (MgSO₄);
- d. 600℃: Magnesium sulfate, being “anhydrous”, then breaks down into components like MgO, SO₃, and SO₂.
Typical Chemical Reactions:
- Reaction with alkalis: Alkali hydroxides such as sodium hydroxide and potassium hydroxide form a flocculent white precipitate of magnesium hydroxide, which is formed due to the - displacement of the sulfate ions by hydroxide ions (MgSO₄ + 2NaOH → Mg(OH)₂↓ + Na₂SO₄);
- Reaction with soluble salts: Barium chloride causes the formation of barium sulfate (BaSO₄) used in qualitative determinations of sulfate ions. Magnahesis reacts with carbonate ions in the solution producing solid magnesium carbonate, MgCO₃ precipitate in the solution;
- Acid, such as hydrochloric acid (e.g., HCl) reactions: Makes та clears the different disulfide such as magnesium chloride and yellow substances such as sulfuric acid.
1.4 Differences and Transformation Between Different Hydrated Forms
II. Preparation Methods of Magnesium Sulfate Heptahydrate
Industrial Preparation: Mainstream Processes and Applicable Scenarios
Process 1: Magnesite Acidolysis Method
Process 2: Direct Processing Method of Magnesium Sulfate Ore
Process 3: Industrial By-product Recovery Method
The raw material sources of this project use salt lake brine which contains Mg²⁺ and SO₄²⁻ and iron and steel plant pickling wastewater which contains MgSO₄ and power plant desulfurization waste liquid which contains MgSO₄. The main procedures of this process begin with brine and wastewater pretreatment which removes all calcium and sodium and heavy metals. The process flow begins with brine and wastewater pretreatment which removes all calcium and sodium and heavy metals. The process continues with evaporation to concentrate the solution which further leads to fractional crystallization. The process starts with sodium chloride crystallization which occurs first before magnesium sulfate heptahydrate begins to form. Chemical industrial parks and enterprises around salt lakes all benefit from this technology. The technology provides strong environmental protection because it executes waste treatment through waste processing. The product achieves 97% to 99% purity while its production cost remains 15% to 20% cheaper than the magnesite production method.
Laboratory Preparation: Small-batch Preparation and Operation Details
3. Process Comparison and Dehydration Reaction: Core Differences Between Different Methods
4. Natural Sources and Derivatives
5. By-product Treatment and Environmental Protection Requirements for Magnesium Sulfate Heptahydrate
III. Application Fields of Magnesium Sulfate Heptahydrate
1. Agricultural Field: Magnesium Sulfate Fertilizer and Feed Additive
Core Application: Agricultural Magnesium Fertilizer
Magnesium sulfate heptahydrate serves as a vital dual-element fertilizer.
- Mechanism of Action: Magnesium acts as a fundamental element for chlorophyll production while enabling plants to perform photosynthesis. Sulfur exists in multiple amino acids and enzymes because it needs to support protein creation and boost crop quality by raising fruit sugar content.
- Applicable Scenarios: Magnesium-deficient soils include acidic red soils and sandy soils.
- Suitable Crops: The plant shows major yield improvements through cultivation of citrus and grapes and tomatoes and rice and soybeans and potatoes and roses.
- Application Methods: The product can be used through foliar spray at 0.2%-0.5% solution and through soil base application at 5-20kg of granular product per mu.
- Precautions: The specific dosage needs to be determined through soil test results since different crops and soil conditions require different adjustments. Excessive use may lead to salt damage or nutrient imbalance.
2. Pharmaceutical Field
Pharmaceutical researchers use magnesium sulfate heptahydrate as a magnesium supplement because its "mild effect and multiple administration routes" make it suitable for use as a treatment method in medical practice.
- Laxative: After oral administration, it increases the osmotic pressure in the intestines, retains water in the intestinal lumen, softens the stool, and stimulates intestinal peristalsis, which is used for treating constipation and intestinal preparation.
- Choleretic: The drug induces gallbladder contraction and bile secretion through its action on the duodenal mucosa, which medical professionals use to treat cholecystitis and gallstones.
- Anticonvulsant: Medical practitioners use intravenous administration to treat eclampsia and tetanus through its central nervous system inhibitory effects, which prevent and control seizures.
- External Anti-swelling Drug: A 50% concentrated injection, when diluted for wet compress, can be used for treating inflammatory swelling, phlebitis, etc.
3. Industrial Field: Functional Materials and Industry Applications
- The Paper Industry uses the material as a "sizing agent" that works with rosin to enhance paper water resistance and as a "filler" which increases both the brightness and texture of paper sheets. The material serves the manufacturing needs of cultural paper and packaging paper production. The standard for the industry can be found in Magnesium Sulfate for Papermaking QB/T 4446-2012.
- The Textile Dyeing Industry uses the substance as a "mordant" to improve dye adhesion on cotton and linen fabrics which prevents color fading and it also serves as a "fixing agent" to enhance printed fabric brightness.
- The material combines with polyvinyl chloride PVC and acrylate resin and epoxy resin and unsaturated polyester and polyurethane to function as an "inorganic flame retardant" The material emits water of crystallization during combustion which absorbs heat and blocks temperature increases while creating an MgO coating that prevents oxygen entry. The product works effectively for wires and cables and building boards.
- The coating industry uses the material as an "extender pigment" which improves latex paint coating performance and scrub resistance through its application. The material functions as an "anti-rust pigment" which metal primers use to prevent metal corrosion.
- Food Additive: The product meets the requirements of Food Additive - Magnesium Sulfate (GB 25588-2010) which establishes regulations for using magnesium sulfate as a "stabilizer" (e.g., in ice cream, dairy products) and a "coagulant" (e.g., in tofu production). The product requires MgSO₄ composition of 99.0% or higher and arsenic content of 0.0002% or lower. The substance is allowed for use in China with a maximum dosage of 3-7g/kg for dairy products and 1.4-2.8g/kg for beverages and milk drinks and 0.05g/kg for mineral drinks.
- The leather industry uses magnesium sulfate heptahydrate as a filler which serves as a reinforcing agent and processing aid to enhance leather physical properties and processing quality.
- The building materials industry uses the material to strengthen building materials while increasing their fire resistance and operational capabilities. The material forms high-performance building materials through its combination with cement and gypsum and other composite materials.
The electroplating industry employs magnesium sulfate as a conductive salt which functions as a plating bath additive to enhance plating layer quality and process efficiency.
4. Daily Life and Other Fields
IV. Comparison and Correlation: Differences from Similar Substances
1. Differences from Common Salts
Magnesium Sulfate Heptahydrate vs. Sodium Carbonate (Na₂CO₃)
Magnesium Sulfate Heptahydrate vs. Magnesium Chloride Hexahydrate (MgCl₂·6H₂O)
Magnesium Sulfate Heptahydrate vs. Epsom Salt
Magnesium Sulfate vs. Magnesium Sulfate Heptahydrate
2. Comparison of Agricultural Magnesium Fertilizers (Magnesium Sulfate Heptahydrate vs. Magnesium Oxide vs. Magnesium Chloride)
3. Environmental Friendliness Comparison (vs. Zinc Sulfate, Calcium Sulfate)
V. Safety and Storage
Risk Control and Operation Specifications
1. Safety and Hazard Identification
2. First Aid Measures
3. Operation and Storage Specifications
4. Emergency Handling and Waste Liquid Treatment
VI. Market and Development: Scale, Enterprises, and Trends
1. Market Scale and Driving Factors
2. Major Manufacturers and Competitiveness
3. Technological Trends and Industry Opportunities & Challenges
VII. Market Quotation and Price-influencing Factors
VIII. Industry Standards: Pharmacopoeias and Industrial Specifications Worldwide
1. Pharmacopoeia Standards (Key Indicators)
2. National Standards for Industrial Products
Chinese GB Standards:
- Industrial Magnesium Sulfate (GB/T 26568-2011): Agricultural-grade MgSO₄·7H₂O ≥98.0%, water-insoluble substances ≤0.2%, pH (5% solution) 5.0-9.0; industrial-grade MgSO₄ ≥99.0%, water-insoluble substances ≤0.1%;
- Feed Additive - Magnesium Sulfate (GB/T 26451-2011): MgSO₄·7H₂O ≥98.0%, heavy metal (Pb) ≤0.001%, arsenic ≤0.0002%;
- Food Additive - Magnesium Sulfate (GB 25588-2010): MgSO₄ ≥99.0%, lead ≤0.0005%, mercury ≤0.0001%.
International Standard: ISO 8298-2000 Magnesium sulfate for industrial use, requiring industrial-grade MgSO₄ ≥98.0% and water-insoluble substances ≤0.5%.
3. EU REACH Regulation Compliance Key Points
IX. Purity Grades
1. Classification by Application and Purity
2. Classification by Form and Particle Size
X. Identification and Quality Control: Methods and Indicators
1. Identification Methods (Laboratory and On-site Rapid Identification)
2. Common Impurities and Their Impacts
3. Quality Standards and Procurement Indicators
XI. Packaging Options: Specifications and Applicable Scenarios
XII. Laizhou Baiyexin Chemical Co., Ltd.: Analysis of Quality and Services
1. Quality Assurance and Certification
2. Core Advantages
3. Production and Service Support
High-quality Production: Features utilization of a complete automation line incorporated with DCS system thus minimizing human efforts and improves the stability of the production cycle (with a limit on the fluctuation of purity at 0.2%). In addition, it employs a dedicated pharmaceutical grade clean workshop with cleanliness level ten 000, thus preventing cross contamination.
Comprehensive Service Support:
- Pre- Sales support. This includes products samples, test analysis and application technical solutions. For instance, agricultural fertilization guidance or recommendations of the industrial formula used.
- In sale: Monitors the development of the logistics, prepares customs declaration according to the export policy, assists the customer in the process of inspection and acceptance.
- After sale service: in the case of a quality problem with the product, if within 24 hours of the complaint the problem is not solved, steps will be taken back, replacement or delivery of the deficient product decided within 48 hours.
4. Customer Success Cases
XIII. Frequently Asked Questions (FAQ)
Magnesium Sulfate Heptahydrate 1-3mm-BAIYEXIN

Baiyexin is the largest magnesium sulfate heptahydrate manufacturer in China, providing magnesium sulfate heptahydrate 1-3mm.
Product SKU: BYX40346
Product Brand: BAIYEXIN
Product Currency: USD
Product Price: 90
Price Valid Until: 2026-12-31
Product In-Stock: InStock
4.99








BaiYeXin –
Magnesium Sulfate Heptahydrate (MgSO₄·7H₂O) has a very good 1-3mm sealed packaging, which does not absorb moisture during long-term storage, making it very friendly to water-soluble fertilizer companies.